Electrostatics
Electrification by Friction
If a polythene rod is rubbed with cloth, the rod can lift small pieces of paper. This can be explained by stating that the rod becomes charged. A charged object will attract a neutral object until it touches and then it is repelled. It is concluded by the experiment that there are two types of charge. It has been decided that the charge that occurs on the polythene rod when it is rubbed is negative and the charge on a perpex rod when it is rubbed shall be called positive. As with magnetism, like charges repel and unlike charges attract.
Insulators and Conductors
Charge does not build up on metalic rods when they are rubbed. The charge can escape through the body to earth, hence metals are known as conductors. Materials such as ebonite and silica do not allow charge to low in this way and are called insulators. For insulators to be effective they must be kept dry and the air around them must also have a low humidity.
Polythene rod charged by rubbing a woolen cap, rod shows negative charge, the woolen cap shows a positive charge. This implies that positive and negative charges are created in equal quantities by friction. If electrons move away from the wool to the polythene the rod will become negatively charged. The nuclei that remain in the wool have lost electrons and so they become positively charged. The process of rubbing effectively increases the area of contact between the wool and the polythene rod, since transfer of charge occurs when the two materials are in contact.
Charging by Induction
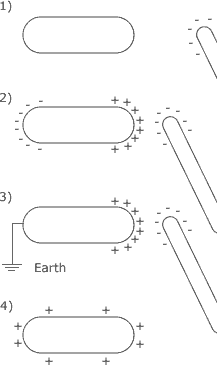
A charged polythene rod can be used to transfer charge onto an insulated conductor without the two object coming into contact. The negatively charged polythene rod is brought close to the conductor. The electrons within the conductor are repelled by the charged polythene rod and the positive ions are attracted to the polythene rod. The conductor has a separation of charge on it. If the negatively charged side is momentarily earthed, the negative charge is able to flow to earth but the positive charge remains attracted to the negatively charged polythene rod. When the rod is taken away from the conductor, the positive charge on the conductor distributes itself evenly around the surface of the conductor leaving it charged.
The Electrophorus
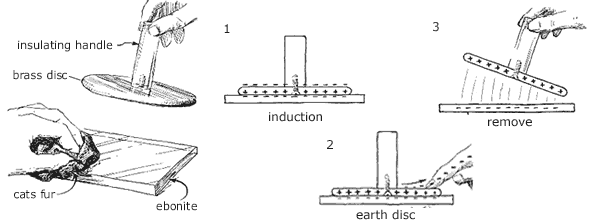
A device that uses this method of charging by induction to carry charge is the electrophorus as shown in Figure 2. The device was invented in 1800 by Volta and consists of an ebonite base which can be charged by rubbing it with catskin which causes the base to become negatively charged. A brass plate with an insulating handle is placed onto the negatively charged base. This causes the electrons in the metal to be repelled and move as far away as possible from the negative charge leaving the positively charge layer attracted to the base. The negative charge is removed by touching the the metal disc briefly, giving a small electric shock to the person as the negative charge flows to earth through the body. The result is a positive charge on the surface of the plate which can be used. The charge on the ebonite plate has not been reduced so the process can in theory be repeated indefinitely.
The Gold-Leaf Electroscope
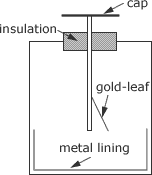
A gold-leaf electroscope is a device that can be used to test for electrical potential and charge and the sign of the charge if it is first given a charge of known sign.
The basic construction consists of an earthed metal box with glass windows, as shown in Figure 3. A metal rod is held in place insulated from the box. On the top of the rod there is a metalic cap and at the other end of rod a strip of gold-leaf is attached part the way up the rod.
When the cap is touched with a charged object, the charge flows down the metal rod and the gold leaf. Since both conductors will have an excess of the same type of charge, the repulsion between like charges causes the gold-leaf to be deflected from the end of the rod.
If the electroscope is first charged with a positive charge and a positive charge is brought near to the cap of the electroscope, the gold leaf will be deflected more because there is a larger potential difference between the leaf and the case. While if a negative charge is brought close to the cap of the electroscope, the gold leaf will fall. No charge will be lost, but the potential difference between the case and the leaf will be reduced.
Faraday's Ice Pail Experiment
Michael Faraday used a metal ice pail as a conducting object to study how charges distribute themeselves when a charged object was placed inside the pail. The experiment, starts with a neutal ice pail, connected to an electroscope. A positively charged sphere is brought over to the metal container. As the charged sphere is moved inside the metal container (but without touching the sides of the ice pail) the sphere induces a negative charge equal to the positive charge on the sphere on the inside of the metal container. On the outside of the metal container, there is also an induced positive charge of equal magnitude as that on the sphere, as indicated by the gold-leaf electroscope.
If the sphere is touched on the inside of the metal container the charge on the sphere flows to the outside of the metal container.
The experiment demonstrates that if a conducting path is provided, charge on any metal object will reside on the outer surface.
If a positively charged sphere is brought close to the outside surface of the pail, the charges will redistribute themselves on the outside of the pail so as to cancel the electric field inside the pail.
Distribution of Potential and Charge over the Surface of a Closed Conductor
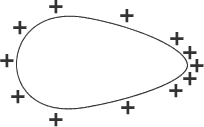
Potential
A pear shaped closed surface conductor as shown in Figure 4. is charged. If wire connected to a gold leaf electroscope is contacted with the surface of the conductor, the electroscope shows a constant deflection. This indicates that the potential on the surface of the conductor is constant all over the surface.
Charge
The charge around the conductor is measured using a proof plane.The proof plane is a small brass disc on an insulated handle. It is first Earthed to ensure there is no charge on the disc. The proof plane is then touched onto the surface of the conductor. The proportion of charge acquired by the disc when it is touched onto the surface of the conduction is proportional to the surface charge density of conductor. The charge can be measured by touching the brass disc of the proof plane on the inside of a metal can standing on the top of the cap of a gold leaf electroscope. The deflection is an indication of the charge at the surface at that point.
If this process is repeated at various positions on the conductor it is found that the least charge is found where the radius of curvature of the conductor is greatest and that the charge is greatest where the surface of the conductor has the smallest radius of curvature, i.e. where it is most pointed. If the radius of curvature is very small, the density of charge can be large enough for charge leakage to occur creating a wind of ions.
